Based in San Diego, Barefoot Solutions is a global agency with 1500+ digital experts trusted by industry leaders.











We design and develop custom applications. With 15 years of experience, we combine the best of cutting edge technology and industry standard best practices.
Nothing is more powerful than a customized AI model tailored to your data and business objectives. Our dedicated machine learning team has the expertise to generate ROI, fast.
Our team of expert marketers works closely with you to develop a custom strategy for success. Our 4 step process includes Consulting, Game-planning, Executing & Optimizing for Growth.



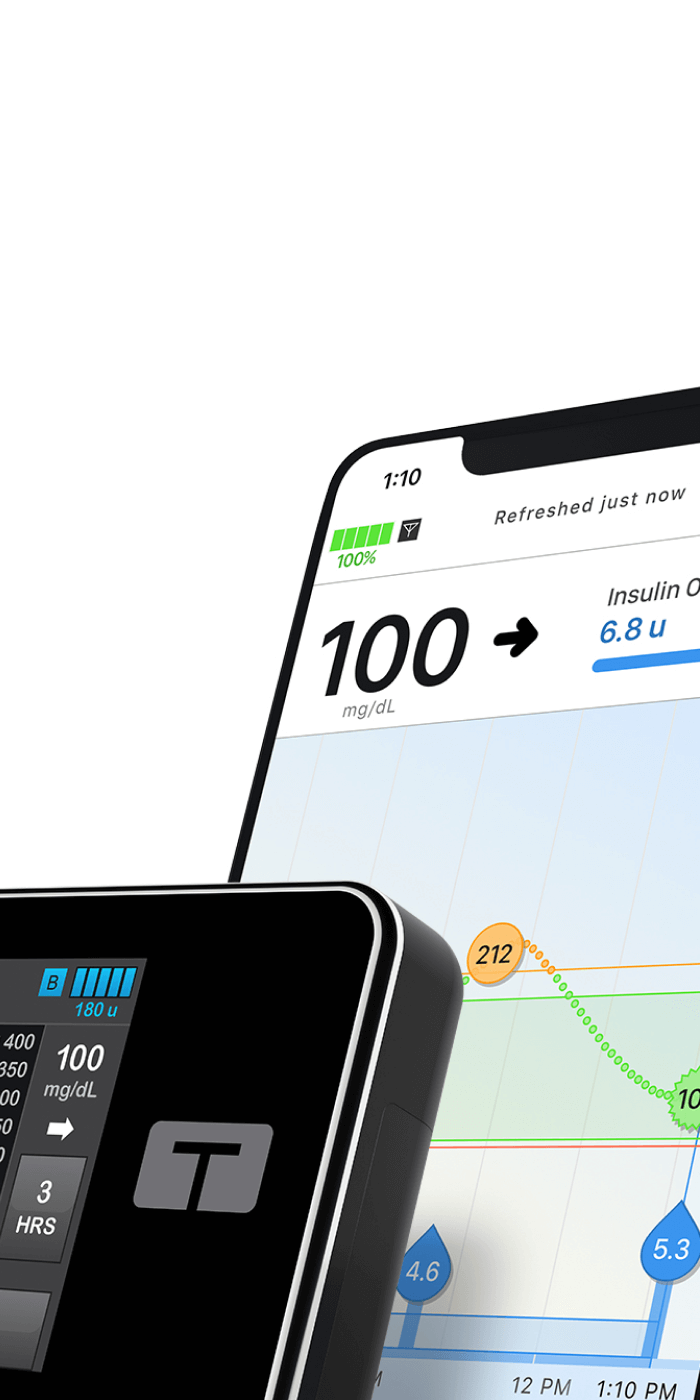
Designing and developing a robust construction management platform, with mobile capability.
SEE CASE STUDY

4241 Jutland Dr., Suite 300
San Diego, CA 92117

“The design looks great, and I have to say that posting an ad is frictionless, nearly instantaneous, and thus pretty easy to do.”

“The app’s entire interface is streamlined, simple to use, and downright slick.”